 Xenon tetrafluoride (XeF4) is a colorless/white crystalline chemical. The most common sort of combustion is fire. What is the combustion process?Ĭombustion is a chemical process in which heat and light are produced. Water may be extracted from a salt solution using this method. What is the process of distillation?ĭistillation is the separation of a mixture of liquids based on variations in their boiling points (or volatility). If the conditions are favorable, this air can continue to rise, cooling and producing Cumulus clouds. As the sun heats the Earth’s surface, the air above it warms and rises. What is convection in the atmosphere?Ĭonvection in the atmosphere is frequently seen in our weather system. Both atoms are involved in the electron transfer from ions. An atom of one element loses one or more electrons during electron transfer, and an atom of another element gains those electrons. Ionic compounds are formed by a process known as electron transfer, in which one atom transfers electrons to another. They also have higher vapor pressures and lower boiling temperatures. They have fewer intermolecular interactions and, as a result, may be transferred to the vapor phase quickly. The term “volatile” refers to compounds with a high evaporation capability. Gaseous SO3 is the primary precursor to acid rain. It is solid at just below room temperature and has a relatively narrow liquid range. It is available in three forms: gaseous monomer, crystalline trimer, and solid polymer. SO3 (sulfur trioxide) is a chemical compound. 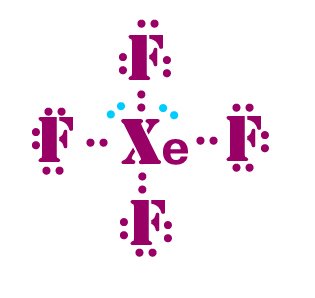
While SF6 is non-toxic when used properly, toxic byproducts can be produced during electrical discharges within SF6-filled equipment, posing a threat to the health of workers who come into contact with them. Sulfur hexafluoride (SF6) is a non-toxic gas that is used in a variety of applications due to its inert properties. This molecule has two lone pairs and three bound pairs, according to the ClF3 Lewis structure. What is CLF3 molecular geometry?ĬlF3 has a T-shaped molecular geometry and trigonal bipyramidal electron geometry. Sulfur electronic configuration is 1s2 2s2 2p6 3s2 3p4 4. With an annual output of 6 million tonnes, decahydrate is an important commodity chemical product. It exists in all forms as white solids that are very water-soluble. It is mostly utilized in the production of detergents and in the Kraft process of paper pulping. It has the formula Na2SO4 and is an inorganic chemical. The sodium salt of sulfuric acid is sodium sulfate. The electrons that are involved in a bond are known as electrons. Sigma bonds are the strongest covalent bonds because the relevant orbitals are directly overlapping. Electrons are the electrons that are involved in forming a connection. Atomic orbitals colliding head-on produce them. In chemistry, sigma bonds (bonds) are the strongest type of covalent chemical bond. Related Links N 2O Lewis Structure| Laughing Gas CO 2 Lewis Structure and Molecular Geometry SO 2 (Sulfur Dioxide) Lewis structure SiO 2 Lewis Structure| Step By Step Construction Frequently Asked Questions (FAQs) 1. Although the individual Xe-F atoms are polar, the Xenon Tetrafluoride molecule is nonpolar because they cancel each other out, resulting in a net dipole moment of zero. 
The central atom, Xenon, is chemically bonded with four fluorine atoms to form a square planar molecular geometry. This is why the compound Xenon tetrafluoride has a square planar structure. To minimize repulsion, the axial positions of the geometry in an octahedral shape are occupied by the lone pairs. The structure of Xenon tetrafluoride is square planar and is made up of non-bonding electrons that form an octahedral shape. Nonbonding electrons are the two lone pairs of electrons, on the other hand. The central Xe atom contains twelve atoms, eight of which share four fluorine bonds. Because XeF4 leaves a residue of metal impurities, it is widely used to degrade silicone rubber. XeF4 is a chemical compound made up of Xenon (Xe) and fluorine atoms. Xenon tetrafluoride FormulaĪ linear molecule with two covalent Xe-F bonds XeF4 can be used to detect and analyze trace metals that contaminate silicone rubber. It is composed of xenon (a noble gas) and fluoride (a naturally occurring mineral). 
Xenon tetrafluoride (XeF4) is a crystalline compound that is normally colorless or white. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
